Getting a generic drug onto the market in Europe isn't as simple as filing one piece of paperwork. It's more like navigating a maze with four different entrances, each with its own set of rules and timelines. For years, this system was a bit of a patchwork, but the EU Pharma Package is a sweeping set of regulatory reforms finalized in June 2025 designed to modernize how medicines are approved and accessed across the Union.
If you're a manufacturer or a healthcare professional, you've probably noticed that generics make up about 65% of prescriptions in the EU by volume. Yet, they only account for 18% of the total value. This gap exists because the regulatory hurdles are high, and the timing of when a generic can actually enter the market is often delayed by national red tape. On average, generics have been delayed by over 11 months across different member states. The 2025 reforms aim to kill that lag and get affordable medicine to patients faster.
Choosing the Right Approval Path
Depending on your budget and which countries you're targeting, you have to pick one of four authorization pathways. Each has a different trade-off between cost, speed, and reach.
First, there's the Centralized Procedure (CP). This is the "gold standard" for big players. You submit one application to the European Medicines Agency (EMA), and if approved, you can sell your drug in all 27 EU member states plus Iceland, Liechtenstein, and Norway. Under the 2025 rules, the scientific assessment time has dropped from 210 to 180 days. It's fast and broad, but it's expensive-costing upwards of €1.6 million when you factor in consultancy fees.
Then you have the Mutual Recognition Procedure (MRP). Here, you get approval in one "Reference Member State" first, and then other countries recognize that approval. It's cheaper than the CP, but it's risky. We've seen cases where pricing disputes in Germany delayed entry into the Netherlands and Belgium by over eight months, even though the drug was technically approved.
The Decentralized Procedure (DCP) lets you apply to several countries at once without having a prior national approval. It's popular, but it's often a headache. About 37% of these applications face delays of six months or more because different countries-especially in Eastern Europe-interpret quality requirements differently.
Finally, the National Procedure is for those who only want to target one specific market. It's the simplest path but offers zero leverage across the rest of the EU.
| Pathway | Scope | Avg. Timeline | Est. Initial Cost | Best For... |
|---|---|---|---|---|
| Centralized (CP) | All EU/EEA | ~226 Days | High (€1.2M+) | High-value, blockbusters |
| Mutual Recognition (MRP) | Selected States | ~133 Days | Medium (€200k) | Fast entry in key hubs |
| Decentralized (DCP) | Selected States | ~247 Days | Medium | Simultaneous multi-market |
| National | One State | 180-240 Days | Low | Niche or single-market focus |
The Technical Bar: Bioequivalence and Quality
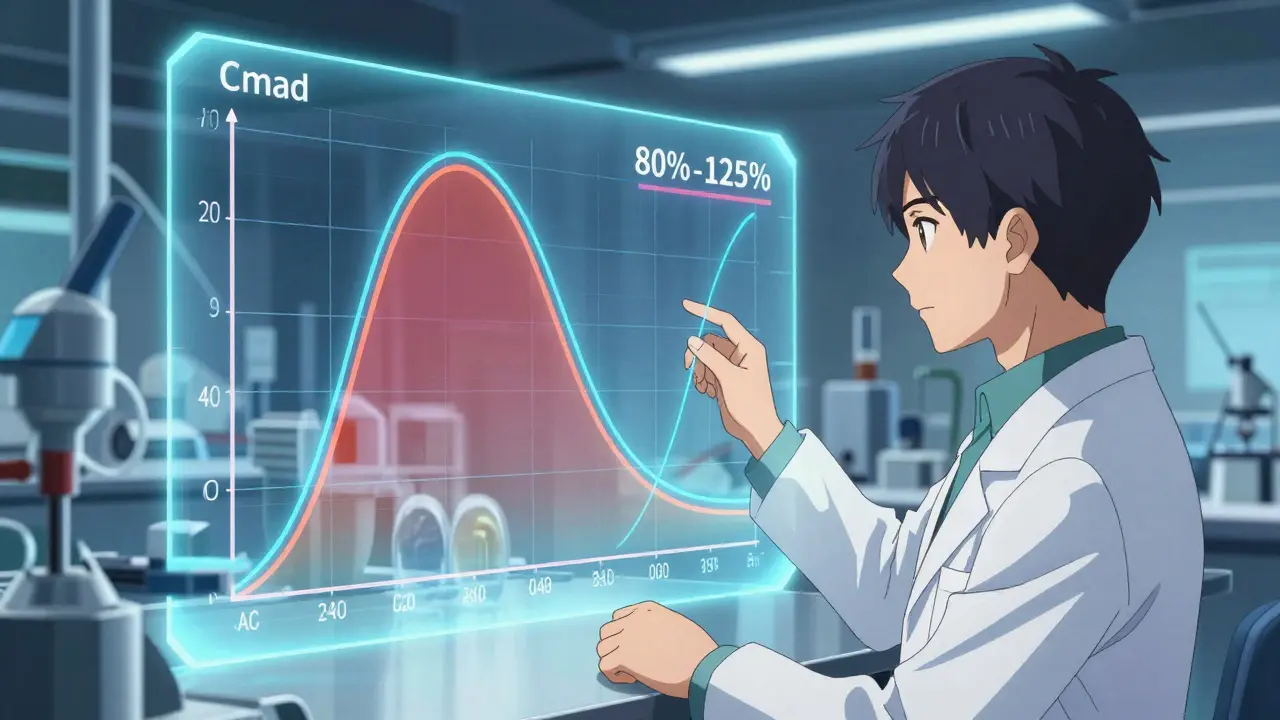
No matter which path you take, you can't just say your drug is "similar." You have to prove bioequivalence. This means showing that your generic version delivers the active ingredient into the bloodstream at the same rate and to the same extent as the original reference product.
The EMA is strict about the math here. Specifically, the 90% confidence intervals for the peak concentration (Cmax) and the total exposure (AUC) must fall between 80% and 125%. If you're outside that window, you're back to the drawing board. For complex generics, like inhalers, the bar is even higher. Germany's BfArM often demands extra pharmacodynamic studies that go beyond the standard EMA requirements, which is a common point of frustration for manufacturers.
Another hurdle is stability data. If your drug contains polymorphic compounds, German authorities will want more data than others. Meanwhile, if you're targeting France, you'll need specific pediatric formulation paperwork to satisfy the ANSM. It's these small, national differences that create the "fragmentation" experts complain about.
How the 2025 Pharma Package Changes the Game
The 2025 reforms aren't just about paperwork; they're about timing and money. One of the biggest wins for generics is the expanded Bolar exemption. In the past, companies could only start pricing and reimbursement talks two months before a patent expired. Now, that window has opened to six months.
Why does this matter? Because it allows generic firms to align their pricing with health technology assessments (HTA) much earlier. Experts predict this could shave over four months off the time it takes to get a drug on the shelf and potentially lower launch prices by up to 18% because of earlier competitive pressure.
There's also a major shift in Regulatory Data Protection (RDP). Starting July 1, 2026, the standard protection period is dropping from 10 years to 9 years (an 8+1 model). While this can be extended back to 10 if certain public health targets are met, the general trend is toward faster generic entry. This change alone could accelerate the launch of nearly 80 high-value biologics currently in the pipeline.

Operational Realities and Digital Shifts
If you're running a regulatory team, your to-do list just got longer. The EU is moving toward mandatory electronic product information (ePI). By 2026, all product info must be submitted in XML format. For a mid-sized company, setting up the IT infrastructure to handle this can cost between €180,000 and €250,000.
Then there's the "obligation to supply." The Critical Medicines Act of March 2025 now requires mandatory stockpiling for 200 essential generics. While this is great for preventing shortages, it adds a layer of cost and new quality verification protocols that can act as a barrier to entry for smaller firms.
We're also seeing a shift in who is winning these markets. Indian manufacturers now hold about 38% of EU generic approvals, up from 29% just a few years ago. However, European giants like Sandoz are fighting back by using the Centralized Procedure to launch EU-wide simultaneously, sometimes beating the competition by nearly a year.
What is the main difference between CP and MRP for generics?
The Centralized Procedure (CP) gives you a single authorization valid across all EU/EEA states, making it ideal for high-value drugs. The Mutual Recognition Procedure (MRP) requires approval in one state first, which is then recognized by others; it's cheaper but carries a higher risk of national delays during the recognition phase.
How does the Bolar exemption expansion affect market entry?
It extends the window for pricing and reimbursement negotiations from 2 months to 6 months before patent expiry. This allows generics to enter the market an average of 4.3 months faster after the patent ends.
What are the bioequivalence requirements for EU generics?
Generics must prove they are therapeutically equivalent to the reference product. Specifically, the 90% confidence intervals for Cmax (peak concentration) and AUC (area under the curve) must fall between 80.00% and 125.00%.
When does the new Regulatory Data Protection (RDP) framework start?
The revised RDP framework, which reduces standard protection to 8 years plus 1 year of market protection, becomes operational on July 1, 2026.
Does the EU require specific formats for product information?
Yes. By 2026, the EU Pharma Package mandates that all product information be submitted as electronic product information (ePI) using XML format.
Next Steps for Manufacturers
If you're planning a launch for 2026 or 2027, start your preparation at least 18 months in advance. Focus on the updated 2025 Bioequivalence Guidelines and ensure your IT team is ready for the XML ePI transition. If you're targeting a high-value biologic, keep a close eye on the July 2026 RDP shift-that's your window to move aggressively. For those targeting smaller markets, be wary of the "obligation to supply" stockpiling rules, as these will require more liquid capital up front than previous cycles.


13 Responses
Absolute chaos! The sheer audacity of the EU to think they can just "modernize" a system that has been a bureaucratic nightmare for decades with a few XML files and a slightly shifted timeline is simply breathtaking. I mean, honestly, who actually believes that a 30-day reduction in assessment time for the Centralized Procedure is going to magically solve the systemic dysfunction of European regulatory bodies? It's a drop in the bucket! We are talking about a continent where the right hand doesn't even know what the left hand is doing half the time, and yet they expect us to be thrilled about a 9-year protection window. It's an absolute travesty for innovation, a slap in the face to the real architects of medicine, and quite frankly, a comedic attempt at reform that only a bureaucrat could love. The drama of these "reforms" is just a smoke screen for their own incompetence!
eu just wantin more money’s thats all it is lol
Typical globalist power grab. Notice how Indian manufacturers are suddenly taking over a huge chunk of the market? It's all designed to dismantle any remaining national sovereignty over health. The US doesn't play these games because we actually understand how to drive innovation without needing a committee of 27 different countries to agree on a font size for an XML document. This whole "Pharma Package" is just a way to standardize the decline of Western medicine while outsourcing the actual production to whoever is cheapest. Wake up people, the centralization of drug approval is just the first step toward a total regulatory monopoly that'll make the FDA look like a lemonade stand.
Exactly what I've been saying!! 🙄 The stockpiling rules are just a front for controlling the supply chain. They want to know exactly who has what so they can flip the switch when they decide we've had enough "essential" meds. It's all a game to them 🤡
The Bolar exemption shift is actually a brilliant little tweak for the underdogs. By opening that window to six months, the smaller labs can actually breathe and get their ducks in a row before the patent cliff hits. It's like getting a head start in a race where the big pharma giants usually have a ten-mile lead. I've seen a few boutique firms in the US try similar timing strategies, and it usually results in a much more vibrant, competitive market once the generics finally drop. It'll be a wild ride for the 2026 biologics pipeline!
It's interesting to see how different regions handle the balance between affordability and innovation.
maybe the world just needs to share more and fight less over who gets the money first we all want to be healthy so why not make it easy for everyone to get the medicine they need without all the rules
Wait... €1.6M for one application???!! That is insane!!! Who even pays that??!!
India is dominating because we have the best talent and the strongest will. EU rules are just hurdles for those who are too slow to adapt. The 38% share is just the beginning of the new era.
I truly feel that we can look at this from a perspective of collective growth, because while the financial burdens of the XML transition might seem daunting for a mid-sized company, the eventual transition to a digital standard could actually foster a more transparent relationship between the regulators and the manufacturers, potentially leading to a world where the patient's needs are finally placed above the rigid adherence to bureaucratic tradition, although we must admit that the road to 2026 will be paved with a lot of frustration and late-night IT troubleshooting for many hardworking teams across the globe.
From an outside perspective, the fragmentation between the German BfArM and the French ANSM seems like a classic example of how national identity persists even within a unified economic bloc. It is quite fascinating to observe how a simple requirement for pediatric formulation in France can create such a significant operational bottleneck for a manufacturer who has already cleared the EMA's hurdles. I wonder if this lingering regionalism is actually a subconscious defense mechanism against the perceived sterilization of national healthcare standards by a centralized European authority.
This whole thing is just a joke for the people who think they're saving the world. It's just shifting money from one corporate pocket to another while pretending the "patient" is the one winning. Please.
lol the IT cost is the best part honestly just imagine the panic when the deadline hits and the XML doesnt work its gonna be a total bloodbath for the small firms